The left atrial appendage dangles off the left atrial chamber in the heart and has long been suspected of being a nidus for clot that can break off and cause a stroke. Patients diagnosed with an arrhythmia called atrial fibrillation have particularly sluggish flow noted in the left atrial appendage and imaging studies corroborate the presence of clots in the left atrial appendage. The treatment of patients with atrial fibrillation has always involved powerful blood thinners to prevent formation of clot. This presents a problem for patients who cannot be anticoagulated, usually because they have demonstrated a propensity for severe bleeding on blood thinners. In a prior age, there were no good options for these patients, and doctors and families were reduced to prayer to prevent strokes from occurring.
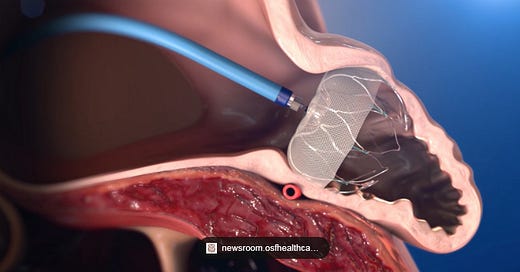
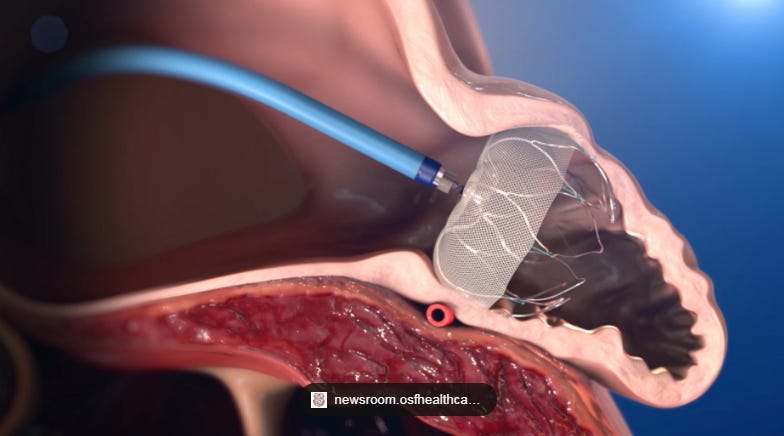
In this context, the arrival of the left atrial appendage occluders which were devices to plug the left atrial appendage and thus abolish the source of the clots seemed to be a welcome development to offer to patients.
The trial that led to the approval of the WATCHMAN device was the PREVAIL trial, a study that randomized high risk patients to Coumadin (a blood thinner) or the WATCHMAN device and observed whether rates of major bleeding or stroke were any different between the two arms. Not surprisingly the group that did not receive a blood thinner (coumadin) had lower rates of bleeding. Surprisingly, this initial trial, did show more strokes in patients who had the WATCHMAN device placed - there were 13 strokes in the patient group assigned to the WATCHMAN device, and only one stroke in patients on coumadin.
The community of WATCHMAN implanters has responded to this data by suggesting the higher stroke risk may be a play of chance, and that as familiarity with the device grows over time, the procedure would get safer over time. But contemporary studies that evaluate for Device Related Thrombus (DRT) raise concerns.
The recently completed Swiss-Apero trial compared the WATCHMAN to a different closure device, and as part of the protocol patients had imaging studies at baseline, 45 days, and one year. Reflecting contemporary practice, most patients at 45 days after implant were either on single antiplatelet or dual antiplatelet therapy. At one year, 10% of patients in both arms were dead, and 9.9% of patients in the WATCHMAN arm had evidence of a definite or probably Device Related Thrombosis (DRT). This should be of great interest to the reader because this annual rate of iatrogenic thrombus formation is higher than the vast majority of all patients with atrial fibrillation walking the earth.
Critics may try to disentangle the risk of stroke from DRT, but the rates of DRT seen, in current practice provide a plausible mechanism for the higher ischemic stroke risk that has been identified since the inception of left atrial appendage occluders. It is easy to find systemic reviews and meta-analyses that seem to suggest the devices are safer than what I’ve presented, but these analyses suffer from a lack of appropriate control arms and are difficult to parse.
Patients and generalist physicians should be particularly wary of those who would make the decision to implant this device seem to be a simple one.
Thrombus attached to a WATCHMAN device.
Anish Koka is a cardiologist in Philadelphia.




Terrific, to the point post.
This was very interesting!